Two closed bulbs of equal volume V containing an ideal gas initially at pressure Pi and temperature T1 are connected through a narrow tube of negligible volume as shown in the figure
By A Mystery Man Writer
Description
Two closed bulbs of equal volume V containing an ideal gas initially at pressure Pi and temperature T1 are connected through a narrow tube of negligible volume as shown in the figure below. The temperature of one of the bulbs is then raised to T2. The final pressure pf is :
Two closed bulbs of equal volume V containing an ideal gas initially at pressure Pi and temperature T1 are connected through a narrow tube of negligible volume as shown in the figure below- The temperature of one of the bulbs is then raised to T2- The final pressure pf is -
Since the above system is a closed one, the total number of moles of the ideal gas will be equal before and after the temperature increase. Hence in the given c
Two closed bulbs of equal volume V containing an ideal gas initially at pressure Pi and temperature T1 are connected through a narrow tube of negligible volume as shown in the figure below- The temperature of one of the bulbs is then raised to T2- The final pressure pf is -
Since the above system is a closed one, the total number of moles of the ideal gas will be equal before and after the temperature increase. Hence in the given c
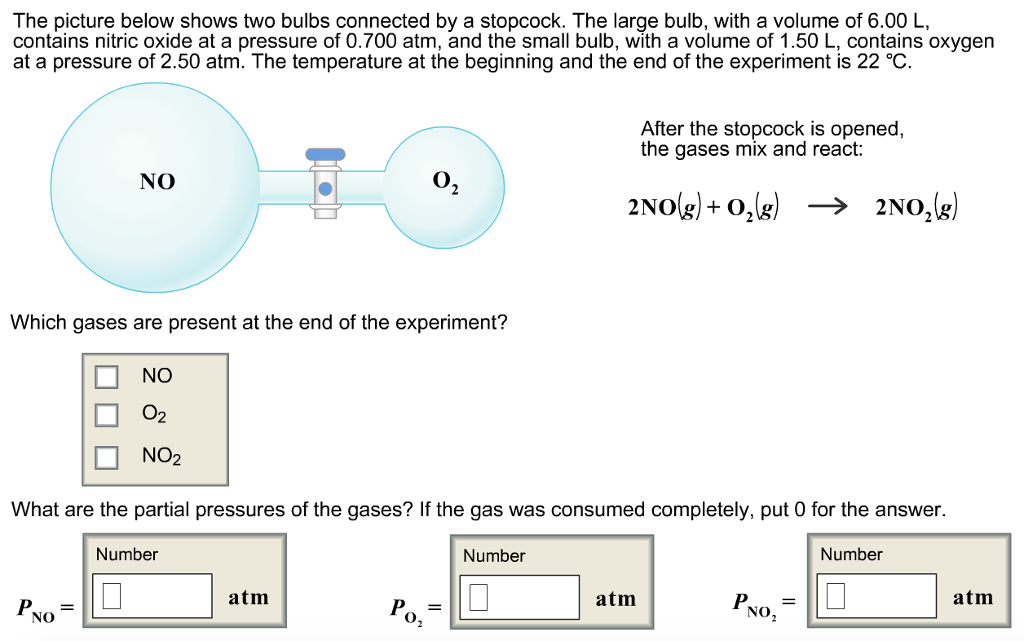
Solved The picture below shows two bulbs connected by a

If a volume containing gas is compressed to half, how many moles of ga

Two closed vessels of equal volume containing air at pressure P(1) and

IIT-JEE Mains 2016 Offline Previous Question Paper Set E

⏩SOLVED:Two closed bulbs of equal volume ( V ) containing an ideal…

Two closed bulbs of equal volume (V) containing an ideal gas initially at pressure p_i and temper

Consider an electrical circuit containing a two way switch 'S'. Initially S is open and then T1 is connected to T2. As the current in R=6 Ω attains a maximum value of
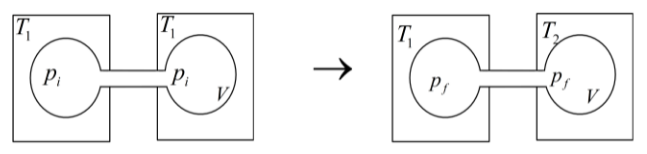
Two closed bulbs of equal volume (\[V\]) containing an ideal gas initially at pressure \[{p_i}\] and temperature \[{T_1}\] are connected through a narrow tube of negligible volume as shown in the figure


If the rms speed of nitrogen at a certain temperature is 3000 ms^(-1)

Solved 10. In an experiment, you have two bulbs connected by
from
per adult (price varies by group size)